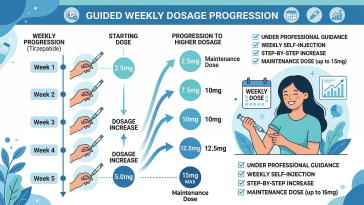
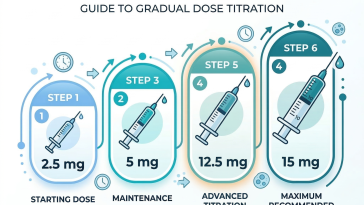
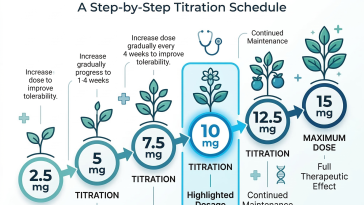
Introduction
Mounjaro (tirzepatide) has gained global attention as a prescription medication used in the management of type 2 diabetes and metabolic health. As awareness of modern treatments grows around the world—including among healthcare discussions in Ghana—it is important for patients to understand how medications should be stored, handled, and verified for safety.
Because prescription medicines require proper manufacturing and storage conditions, patients should always obtain them through licensed healthcare providers or regulated medical channels.
Why Medication Authenticity Matters
Like many prescription medicines, tirzepatide must be manufactured, transported, and stored according to strict pharmaceutical standards. Improper storage or handling can affect the safety and effectiveness of a medication.
Healthcare professionals emphasize that patients should always ensure that medicines come from regulated and trustworthy medical sources.
How to Recognize Authentic Manufacturer Packaging
Original manufacturer packaging typically includes:
-
sealed outer packaging
-
clearly printed batch and expiry information
-
patient information leaflet included in the box
-
consistent and high-quality labeling
If packaging appears damaged, altered, or unclear, patients should consult a pharmacist or healthcare professional before using the medication.
Proper Storage Requirements
Temperature-sensitive medicines such as tirzepatide require appropriate storage conditions.
Typical storage guidance may include:
-
refrigeration between 2°C and 8°C
-
avoiding freezing temperatures
-
protecting the medication from direct sunlight
-
maintaining sealed packaging until use
Patients should follow the storage instructions provided by healthcare professionals or printed in the medication leaflet.
Checking the Injection Pen
Injection devices manufactured for prescription medications usually have:
-
clear labeling and dosage markings
-
intact protective caps
-
a sealed design without leaks or damage
-
smooth mechanical operation
If a device appears damaged or tampered with, it should not be used until it has been reviewed by a healthcare professional.
Why Proper Handling Is Important
Medicines that are not stored or handled correctly may lose effectiveness or pose potential health risks. Healthcare providers recommend obtaining medications through licensed medical channels where quality control standards are maintained.
Patients should always seek guidance from a qualified healthcare professional when starting or using any prescription medication.
Safe Storage at Home
If prescribed tirzepatide, patients are usually advised to follow general medication safety practices such as:
-
keeping the medication refrigerated within recommended temperature ranges
-
avoiding exposure to heat or sunlight
-
keeping medication out of reach of children
-
following instructions provided by a healthcare professional
Always consult the medication leaflet or healthcare provider for specific storage recommendations.
Final Advice
Understanding how prescription medications should be packaged, stored, and handled is an important part of maintaining safe and effective treatment. Patients should always consult licensed healthcare professionals when seeking guidance on diabetes or metabolic health medications.
Medical Disclaimer
This article is provided for educational and informational purposes only. It does not promote, sell, or distribute prescription medications.
Mounjaro (tirzepatide) is a prescription medicine that should only be used under the supervision of a licensed healthcare professional. Always consult a qualified medical provider before starting, stopping, or changing any medication.